Plasmodium falciparum
| Plasmodium falciparum | |
|---|---|
 |
|
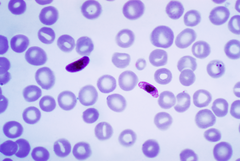
| Blood smear of Plasmodium falciparum (gametocytes - sexual forms) | |
| Scientific classification | |
| Domain: | Eukarya |
| Kingdom: | Chromalveolata |
| Phylum: | Apicomplexa |
| Class: | Aconoidasida |
| Order: | Haemosporida |
| Family: | Plasmodiidae |
| Genus: | Plasmodium |
| Species: | P. falciparum |
| Binomial name | |
| Plasmodium falciparum Welch, 1897 |
|
Plasmodium falciparum is a protozoan parasite, one of the species of Plasmodium that cause malaria in humans. It is transmitted by the female Anopheles mosquito. P. falciparum is the most dangerous of these infections as P. falciparum (or malignant) malaria has the highest rates of complications and mortality. As of 2006 it accounted for 91% of all 247 million human malarial infections (98% in Africa)[1] and 90% of the deaths. It is more prevalent in sub-Saharan Africa than in other regions of the world; in most African countries, more than 75% of cases were due to P.falciparum, whereas in most other countries with malaria transmission, other Plasmodial species predominate.[2]. A map based on the reported incidence of infection in 2007 is available at: http://www.map.ox.ac.uk/
Background
Malaria is caused by an infection with protozoa of the genus Plasmodium. The name malaria, from the Italian mala aria, meaning bad air, comes from the linkage suggested by Giovanni Maria Lancisi (1717) of malaria with the poisonous vapours of swamps. This species name comes from the Latin falx meaning sickle, and parere meaning to give birth. The organism itself was first seen by Laveran on November 6, 1880 at a military hospital in Constantine, Algeria, when he discovered a microgametocyte exflagellating. Manson (1894) hypothesised that mosquitoes could transmit malaria. This hypothesis was experimentally confirmed independently by Giovanni Battista Grassi and Ronald Ross in 1898. Grassi (1900) proposed an exerythrocytic stage in the life cycle, later confirmed by Short, Garnham, Covell and Shute (1948), who found Plasmodium vivax in the human liver.
Around the world, malaria is the most significant parasitic disease of humans and claims the lives of more children worldwide than any other infectious disease. Since 1900, the area of the world exposed to malaria has been halved, yet two billion more people are presently exposed. Morbidity, as well as mortality, is substantial. Infection rates in children in endemic areas are of the order of 50%: Chronic infection has been shown to reduce school scores by up to 15%. Reduction in the incidence of malaria coincides with increased economic output.
While there are no effective vaccines for any of the six or more species that cause human malaria, drugs have been employed for centuries. In 1640, Huan del Vego first employed the tincture of the cinchona bark for treating malaria: The native Indians of Peru and Ecuador had been using it even earlier for treating fevers. Thompson (1650) introduced this "Jesuits' bark" to England: Its first recorded use there was by Dr John Metford of Northampton in 1656. Morton (1696) presented the first detailed description of the clinical picture of malaria and of its treatment with cinchona. Gize (1816) studied the extraction of crystalline quinine from the cinchona bark and Pelletier and Caventou (1820) in France extracted pure quinine alkaloids, which they named quinine and cinchonine.
Plasmodium life cycle
The life cycle of all Plasmodium species is complex. Infection in humans begins with the bite of an infected female Anopheline mosquito. Sporozoites released from the salivary glands of the mosquito enter the bloodstream during feeding quickly invade liver cells (hepatocytes). Sporozoites are cleared from the circulation within 30 minutes. During the next 14 days in the case of P. falciparum, the liver-stage parasites differentiate and undergo asexual multiplication resulting in tens of thousands of merozoites which burst from the hepatoctye. Individual merozoites invade red blood cells (erythrocytes) and undergo an additional round of multiplication producing 12-16 merozoites within a schizont. The length of this erythrocytic stage of the parasite life cycle depends on the parasite species: 48 hours for P. falciparum, P. vivax, and P. ovale and 72 hours for P. malariae. The clinical manifestations of malaria, fever and chills, are associated with the synchronous rupture of the infected erythrocyte. The released merozoites go on to invade additional erythrocytes. Not all of the merozoites divide into schizonts, some differentiate into sexual forms, male and female gametocytes. These gametocytes are taken up by a female anophylean mosquito during a blood meal. Within the mosquito midgut, the male gametocyte undergoes a rapid nuclear division, producing 8 flagellated microgametes which fertilize the female macrogamete. The resulting ookinete traverses the mosquito gut wall and encysts on the exterior of the gut wall as a oocyst. Soon the oocyst ruptures, releasing hundreds of sporozoites into the mosquito body cavity where they eventually migrate to the mosquito salivary gland.
Pathogenesis
Plasmodium falciparum causes severe malaria via a distinctive property not shared by any other human malaria, that of sequestration. Within the 48-hour asexual blood stage cycle, the mature forms change the surface properties of infected red blood cells causing them to stick to blood vessels (a process called cytoadherence). This leads to obstruction of the microcirculation and results in dysfunction of multiple organs, typically the brain in cerebral malaria[3].
Microscopic appearance

Among medical professionals, the preferred method to diagnose malaria and determine which species of Plasmodium is causing the infection is by examination of a blood film under microscope in a laboratory. Each species has distinctive physical characteristics that are apparent under a microscope. In P. falciparum, only early (ring-form) trophozoites and gametocytes are seen in the peripheral blood. It is unusual to see mature trophozoites or schizonts in peripheral blood smears as these are usually sequestered in the tissues. The parasitised erythrocytes are not enlarged, and it is common to see cells with more than one parasite within them (multiply parasitised erythrocytes). On occasion, faint comma-shaped red dots called "Maurer's dots" are seen on the red cell surface. The comma-shaped dots can also appear as pear-shaped blotches.
Influence of Plasmodium falciparum on the human genome
The presence of the parasite in human populations caused selection in the human genome in a multitude of ways, as humans have been forced to develop resistance to the disease. Beet, a doctor working in Southern Rhodesia (now Zimbabwe) in 1948, first suggested that sickle-cell disease could offer some protection to malaria. This suggestion was reiterated by J. B. S. Haldane in 1949 who suggested that thalassaemia could provide similar protection. This hypothesis has since been confirmed and has been extended to hemoglobin C and hemoglobin E, abnormalities in ankyrin and spectrin (ovalocytosis , elliptocytosis), in glucose-6-phosphate dehydrogenase deficiency and pyruvate kinase deficiency, loss of the Gerbich antigen (glycophorin C) and the Duffy antigen on the erythrocytes, thalassemias and variations in the major histocompatibility complex classes 1 and 2 and CD32 and CD36.
The Plasmodium falciparum genome
In 1995 a consortium - the malaria genome project (MGP) - was set up to sequence the genome of P. falciparum. The genome of the parasite mitochondrion was reported in 1995, that of the plastid (apicoplast) in 1996, and the sequence of the first nuclear chromosome (Chromosome 2) in 1998. The sequence of chromosome 3 was reported in 1999 and the entire genome on 3 October 2002. Annotated genome data can now be fully analyzed at several database resources including the UCSC Malaria Genome Browser, PlasmoDB and GeneDB. The ~24 megabase genome is extremely AT rich (~80%) and is organised into 14 chromosomes: just over 5300 genes were described.
Plasmodium falciparum and sickle cell anemia
Individuals with sickle cell anemia and individuals with sickle cell trait do have reduced parasitemia when compared to wild-typed individuals for the hemoglobin protein in red blood cells. Studies have shown these genetic deviations of hemoglobin from normal states provide protection against the deadly parasite that causes malaria (Allison, 1954). Of the four malarial parasites, Plasmodium falciparum causes the most fatal and medically severe form. Malaria is prevalent in tropical countries with an incidence of 300 million per year and a mortality rate of 1 to 2 million per year. Roughly 50% of all malarial infections are caused by Plasmodium falciparum (Roberts and Janovy Jr., 2005) Upon infection via a bite from an infected Anopheles mosquito, sporozoites devastate the human body by first infecting the liver. While in the liver, sporozoites undergo asexual development and merozoites are released into the blood stream. The trophozoites further develop and reproduce by invading red blood cells. During the reproduction cycle, Plasmodium falciparum produces up to 40,000 merozoites in one day. Other blood sporozoans, such as Plasmodium vivax, Plasmodium ovale, and Plasmodium malariae, that infect humans and cause malaria do not have such a productive cycle for invasion. The process of bursting red blood cells does not have any symptoms, however destruction of the cells does cause anemia, since the bone marrow cannot compensate for the damage. When red blood cells rupture, hemozoin wastes cause cytokine release, chills, and then fever (Roberts and Janovy Jr., 2005).
Plasmodium falciparum trophozoites develop sticky knobs in red blood cells, which then adhere to endothelial cells in blood vessels, thus evading clearance in the spleen. The acquired adhesive nature of the red blood cells may cause cerebral malaria when sequestered cells prevent oxygenation of the brain. Symptoms of cerebral malaria include impaired consciousness, convulsions, neurological disorder, and coma (Brown University). Additional complications from Plasmodium falciparum induced malaria include advanced immunosuppression (Roberts and Janovy Jr., 2005).
Individuals with sickle cell trait and sickle cell anemia are privileged because they have altered sticky knobs. Research by Cholera, R., Brittain, N., Gillrie, M. et al. (2007) has shown that parasitemia (the ability of a parasite to infect) because merozoites of each parasite species that cause malaria invade the red blood cell in three stages: contact, attachment, and endocytosis. Individuals suffering from sickle cell anemia have deformed red blood cells that interfere with the attachment phase and Plasmodium falciparum and the other forms of malaria have trouble with endocytosis.
These individuals have reduced attachment when compared to red blood cells with the normally-functioning hemoglobin because of differing protein interactions. In normal circumstances, merozoites enter red blood cells through two PfEMP-1 protein-dependent interactions. These interactions promote the malaria inflammatory response associated with symptoms of chills and fever. When these proteins are impaired, as in sickle cell cases, parasites cannot undergo cytoadherance interactions and cannot infect the cells; therefore sickle cell-anemic individuals and individuals carrying the sickle cell trait have lower parasite loads and shorter time for symptoms than individuals expressing normal red blood cells (Mockenhaupt, 2004).
Individuals with sickle cell anemia may also experience greatly reduced symptoms of malaria because Plasmodium falciparum trophozoites cannot bind to hemoglobin in order to form sticky knobs. Without knob binding complexes, which is an exclusive feature of Plasmodium falciparum, red blood cells do not stick to endothelial walls of blood vessels, and infected individuals do not experience symptoms such as cerebral malaria (Cholera, R., Brittain, N., Gillrie, M. et al. 2007).
Many may wonder why natural selection has not phased out sickle cell anemia. The answer lies within answers generated by Cholera, R., Brittain, N., Gillrie, M. et al. (2007). Individuals with sickle cell trait are greatly desired in areas where malarial infections are endemic. Malaria kills between 1 and 2 million people per year. It is the leading cause of death among children in tropical regions. Individuals with sickle cell deformities are able to fight Plasmodium parasite infections and do not become victims of malarial demise. Therefore, individuals expressing the genes and individuals carrying genes are selected to remain within the population (Allison 1964). To no surprise the incidence of sickle cell anemia match endemic regions for malarial infections.
Known vectors
Anopheles gambiae (Principal vector) [4]
Anopheles albimanus
Anopheles freeborni
Anopheles maculatus
Anopheles stephensi
Origins and evolution
The closest relative of Plasmodium falciparum is Plasmodium reichenowi, a parasite of chimpanzees. P. falciparum and P. reichenowi are not closely related to the other Plasmodium species that parasitize humans, or indeed mammals generally. It has been argued that these two species originated from a parasite of birds[5]. More recent analyses do not support this, however, instead suggesting that the ability to parasitize mammals evolved only once within the genus Plasmodium[6].
A third species has been discovered that appears to related to these two: Plasmodium gaboni. This putative species is currently (2009) known only from two DNA sequences and awaits a full species description before it can be regarded as valid.
Molecular clock analyses suggest that P. falciparum is as old as the human line; the two species diverged at the same time as humans and chimpanzees[7]. However, low levels of polymorphism within the P. falciparum genome suggest a much more recent origin[8]. It may be that this discrepancy exists because P. falciparum is old, but its population recently underwent a great expansion[9]. The relationship between these parasites has now been clarified somewhat. It appears that P. reichenowi was the ancestor of P. falciparum [10]. The timing of this event is unclear at present but it has been proposed that it may have occurred about 10,000 years ago.
More recently, P. falciparum has evolved in response to human interventions. Most strains of malaria can be treated with chloroquine, but P. falciparum has developed resistance to this treatment. A combination of quinine and tetracycline has also been used, but there are strains of P. falciparum that have grown resistant to this treatment as well. Different strains of P. falciparum have grown resistant to different treatments. Often the resistance of the strain depends on where it was contracted. Many cases of malaria that come from parts of the Caribbean and west of the Panama Canal as well as the Middle East and Egypt can often be treated with chloroquine, since they have not yet developed resistance. Nearly all cases contracted in Africa, India, and southeast Asia have grown resistant to this medication and there have been cases in Thailand and Cambodia in which the strain has been resistant to nearly all treatments. Often the strain grows resistant to the treatment in areas where the use is not as tightly regulated.
Like most Apicomplexa, malaria parasites harbor a plastid similar to plant chloroplasts, which they probably acquired by engulfing (or being invaded by) a eukaryotic alga, and retaining the algal plastid as a distinctive organelle encased within four membranes (see endosymbiotic theory). The apicomplexan plastid, or apicoplast, is an essential organelle, thought to be involved in the synthesis of lipids and several other compounds, and provides an attractive target for antimalarial drug development, particularly in light of the emergence of parasites resistant to chloroquine and other existing antimalarial agents.
Treatment
Uncomplicated falciparum malaria
According to WHO guidelines 2010,[11] artemisinin-based combination therapies (ACTs) are the recommended first line antimalarial treatments for uncomplicated malaria caused by P. falciparum.[11] The following ACTs are recommended by the WHO:[11]
- artemether plus lumefantrine
- artesunate plus amodiaquine
- artesunate plus mefloquine
- artesunate plus sulfadoxine-pyrimethamine
- dihydroartemisinin plus piperaquine
The choice of ACT in a country or region will be based on the level of resistance to the constituents in the combination.[11] Artemisinin and its derivatives should not be used as monotherapy in uncomplicated falciparum malaria.[11] As second-line antimalarial treatment, when initial treatment doesn’t work or stops working, it is recommended to use an alternative ACT known to be effective in the region, such as:
- Artesunate plus tetracycline or doxycycline or clindamycin.[11]
- Quinine plus tetracycline or doxycycline or clindamycin[11]
Any of these combinations are to be given for 7 days.[11]
For pregnant women, the recommended first-line treatment during the first trimester is quinine plus clindamycin for 7 days.[11] Artesunate plus clindamycin for 7 days is indicated if this treatment fails.[11] Still, an ACT is indicated only if this is the only treatment immediately available, or if treatment with 7-day quinine plus clindamycin fails or if there is uncertainty of compliance with a 7-day treatment.[11] In second and third trimesters, the recommended treatment is an ACT known to be effective in the country/region or artesunate plus clindamycin for 7 days, or quinine plus clindamycin for 7 days.[11] Lactating women should receive standard antimalarial treatment (including ACTs) except for dapsone, primaquine and tetracyclines.
In infants and young children, the recommended first-line treatment is ACTs, with attention to accurate dosing and ensuring the administered dose is retained.[11]
For travellers returning to non-endemic countries, any of the following is recommended:[11]
- atovaquone-proguanil
- artemether-lumefantrine;
- quinine plus doxycycline or clindamycin.
Severe falciparum malaria
In severe falciparum malaria, it is recommended that rapid clinical assessment and confirmation of the diagnosis is made, followed by administration of full doses of parenteral antimalarial treatment without delay with whichever effective antimalarial is first available.[11]
For adults, intravenous (IV) or intramuscular (IM) artesunate is recommended.[11] Quinine is an acceptable alternative if parenteral artesunate is not available.[11]
For children, especially in the malaria endemic areas of Africa, any the following antimalarial medicines is recommended:[11]
- artesunate IV or IM
- quinine (IV infusion or divided IM injection);
- artemether IM. It should only be used if none of the alternatives are available as its absorption may be erratic.[11]
Parenteral antimalarials should be administered for a minimum of 24 h in the treatment of severe malaria, irrespective of the patient’s ability to tolerate oral medication earlier.[11] Thereafter, it is recommended to complete treatment by giving a complete course of any of the following:[11]
- an ACT
- artesunate plus clindamycin or doxycycline;
- quinine plus clindamycin or doxycycline.
If complete treatment of severe malaria is not possible, it is recommended that patients are given pre-referral treatment and referred immediately to an appropriate facility for further treatment. The following are options for pre-referral treatment:[11]
- rectal artesunate
- quinine IM
- artesunate IM
- artemether IM.
History of falciparum treatment
Attempts to make synthetic antimalarials began in 1891. Atabrine, developed in 1933, was used widely throughout the Pacific in World War II but was deeply unpopular because of the yellowing of the skin it caused. In the late 1930s, the Germans developed chloroquine, which went into use in the North African campaigns. Mao Zedong encouraged Chinese scientists to find new antimalarials after seeing the casualties in the Vietnam War. Artemisinin was discovered in the 1970s based on a medicine described in China in the year 340. This new drug became known to Western scientists in the late 1980s and early 1990s and is now a standard treatment. In 1976 P. falciparum was successfully cultured in vitro for the first time which facilitated the development of new drugs substantially.[12] A 2008 study published in the New England Journal of Medicine highlighted the emergence of artemisinin-resistant strains of P.falciparum in Cambodia.[13]
Vaccination
Although an antimalarial vaccine is urgently needed, infected individuals never develop a sterilizing (complete) immunity, making the prospects for such a vaccine dim. The parasites live inside cells, where they are largely hidden from the immune response. Infection has a profound effect on the immune system including immune suppression. Dendritic cells suffer a maturation defect following interaction with infected erythrocytes and become unable to induce protective liver-stage immunity. Infected erythrocytes directly adhere to and activate peripheral blood B cells from nonimmune donors. The var gene products, a group of highly expressed surface antigens, bind the Fab and Fc fragments of human immunoglobulins in a fashion similar to protein A to Staphylococcus aureus, which may offer some protection to the parasite from the human immune system. Despite the poor prospects for a fully-protective vaccine, it may be possible to develop a vaccine that would reduce the severity of malaria for children living in endemic areas.
See also
List of parasites (human)
UCSC Malaria Genome Browser
Sources and further reading
Overview
Spatial distribution
Blood slides
- Blood forms
- Blood forms
- Blood forms
- Blood forms
- Multiple blood forms
- Female gametocyte
- Female gametocyte
Case histories
Pathology due to Plasmodium falciparum
- Brain
- Gross pathology
- Low power H & E stain
- High power H & E stain showing parasite adherence to the vessel walls
- Spleen
- Liver
- Kidney
Plasmodium falciparum genome data
- Gardner, MJ; Hall, N; Fung, E; White, O; Berriman, M; Hyman, RW; Carlton, JM; Pain, A et al.; Hall, N.; Fung, E.; White, O.; Berriman, M.; Hyman, R.W.; Carlton, J.M.; Pain, A.; Nelson, K.E.; Bowman, S.; Others, (2002). "Genome sequence of the human malaria parasite Plasmodium falciparum". Nature 419 (6906): 498–511. doi:10.1038/nature01097. PMID 12368864.
- PlasmoDB: The Plasmodium Genome Resource
- GeneDB Plasmodium falciparum
- Genome
- UCSC Plasmodium Falciparum Browser
Other
- Colombian scientists develop computacional tool to detect the plasmodium falciparum (in spanish)
- Allison AC (February 1954). "Protection afforded by sickle-cell trait against subtertian malareal infection". Br Med J 1 (4857): 290–4. doi:10.1136/bmj.1.4857.290. PMID 13115700.
- Allison AC (1964). "Polymorphism and Natural Selection in Human Populations". Cold Spring Harb. Symp. Quant. Biol. 29: 137–49. PMID 14278460.
- Cholera R, Brittain NJ, Gillrie MR, et al. (January 2008). "Impaired cytoadherence of Plasmodium falciparum-infected erythrocytes containing sickle hemoglobin". Proc. Natl. Acad. Sci. U.S.A. 105 (3): 991–6. doi:10.1073/pnas.0711401105. PMID 18192399. PMC 2242681. http://www.pnas.org/cgi/pmidlookup?view=long&pmid=18192399.
- Mockenhaupt FP, Ehrhardt S, Otchwemah R, et al. (May 2004). "Limited influence of haemoglobin variants on Plasmodium falciparum msp1 and msp2 alleles in symptomatic malaria". Trans. R. Soc. Trop. Med. Hyg. 98 (5): 302–10. doi:10.1016/j.trstmh.2003.10.001. PMID 15109555. http://linkinghub.elsevier.com/retrieve/pii/S0035920303000890.
- Janovy, John; Roberts, Larry S. (2004). Foundations of Parasitology (7th ed.). McGraw-Hill Education (ISE Editions). ISBN 0-07-111271-5.
References
- ↑ "World Malaria Report 2008". World Health Organisation. 2008. pp. 10. http://apps.who.int/malaria/wmr2008/malaria2008.pdf. Retrieved 2009-08-17.
- ↑ "World Malaria Report 2008". World Health Organisation. 2008. pp. 11–12. http://apps.who.int/malaria/wmr2008/malaria2008.pdf. Retrieved 2009-08-17.
- ↑ Dondorp AM, Pongponratn E, White NJ (February 2004). "Reduced microcirculatory flow in severe falciparum malaria: pathophysiology and electron-microscopic pathology". Acta Trop. 89 (3): 309–17. doi:10.1016/j.actatropica.2003.10.004. PMID 14744557.
- ↑ Mbogo CM, Mwangangi JM, Nzovu J, et al. (June 2003). "Spatial and temporal heterogeneity of Anopheles mosquitoes and Plasmodium falciparum transmission along the Kenyan coast". Am. J. Trop. Med. Hyg. 68 (6): 734–42. PMID 12887036.
- ↑ Rathore, D; Wahl AM, Sullivan M, McCutchan TF (2001-04-25). "A phylogenetic comparison of gene trees constructed from plastid, mitochondrial and genomic DNA of Plasmodium species". Molecular and Biochemical Parasitology 114 (1): 89–94. doi:10.1016/S0166-6851(01)00241-9. PMID 11356517.
- ↑ Yotoko, KSC; C Elisei (2006-11). "Malaria parasites (Apicomplexa, Haematozoea) and their relationships with their hosts: is there an evolutionary cost for the specialization?". Journal of Zoological Systematics and Evolutionary Research 44 (4): 265–73. doi:10.1111/j.1439-0469.2006.00377.x.
- ↑ Escalante, AA; E Barrio, FJ Ayala (1995). "Evolutionary origin of human and primate malarias: evidence from the circumsporozoite protein gene". Molecular Biology and Evolution 12 (4): 616–26. ISSN 07374038. PMID 7659017.
- ↑ Hartl, DH (2004-01). "The origin of malaria: mixed messages from genetic diversity". Nature Reviews: Microbiology 2 (1): 15–22. doi:10.1038/nrmicro795. PMID 15035005.
- ↑ Joy, DA; Feng X, Mu J, Furuya T, Chotivanich K, Krettli AU, Ho M, Wang A, White NJ, Suh E, Beerli P, Su XZ. (2003-04-11). "Early origin and recent expansion of Plasmodium falciparum". Science 300 (5617): 318–21. doi:10.1126/science.1081449. PMID 12690197.
- ↑ Rich SM, Leendertz FH, Xu G, Lebreton M, Djoko CF, Aminake MN, Takang EE, Diffo JL, Pike BL, Rosenthal BM, Formenty P, Boesch C, Ayala FJ, Wolfe ND (2009). "The origin of malignant malaria". Proc Natl Acad Sci USA 106 (35): 14902–7. doi:10.1073/pnas.0907740106. PMID 19666593.
- ↑ 11.00 11.01 11.02 11.03 11.04 11.05 11.06 11.07 11.08 11.09 11.10 11.11 11.12 11.13 11.14 11.15 11.16 11.17 11.18 11.19 11.20 11.21 Guidelines for the treatment of malaria, second edition Authors: WHO. Number of pages: 194. Publication date: 2010. Languages: English. ISBN: 9789241547925
- ↑ Trager, W; J B Jensen (1976-08-20). "Human malaria parasites in continuous culture". Science (New York, N.Y.) 193 (4254): 673–5. doi:10.1126/science.781840. PMID 781840.
- ↑ Noedl H, Se Y, Schaecher K, Smith BL, Socheat D, Fukuda MM (December 2008). "Evidence of artemisinin-resistant malaria in western Cambodia". N. Engl. J. Med. 359 (24): 2619–20. doi:10.1056/NEJMc0805011. PMID 19064625.
|
||||||||||||||||||||||||||||||||